Biswadip Banerji , Ph.D.
Chief Scientist, & Head of the Department, Central Instrumentation Facility (CIF, IICB)
Organic & Medicinal Chemistry

Research Focus
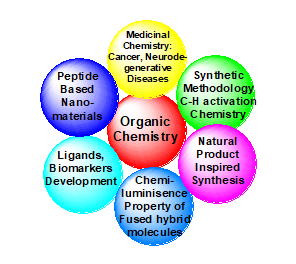
Our research is based on synthetic Organic Chemistry as we love to develop new smethodologies to make new molecules, i.e, ‘New Chemical Entities’ (NCEs). The major objectives remain towards the applications of these newly synthesized molecules. Thus, to our group, ‘Why (?)’ is more important than ‘What to synthesize’. Towards this goal, we follow a simple rule, (i) Selecting an application-oriented target molecular scaffold, (ii) a new synthetic methodology is explored and developed to generate a targeted scaffold library (iii) studies to establish an application of these molecules. The following areas are of the current ‘research interests’ in our laboratory. Overall, our research is open and diverse. Thus, students get the liberty and exposure to explore any application-oriented ideas aligned to the research theme. Students can work on targeted Organic Synthesis/methodologies and explore their applications at the interface of Chemistry & Biology, like Medicinal chemistry (Therapeutics), nano-material chemistry (peptide based), chemo/Bio-sensor (Fluoride/Hydrazine/Cysteine/H2O2 etc detection), bio-markers (cellular organelles like lysosome, mitochondria, nucleus, ER etc), cancer-bio-marker etc. Specific examples: in an ongoing project in the laboratory, we design and synthesize new ‘poly-N-fused heterocycles’ by combining various known bioactive heterocycles. We observed that these fused-heterocyclic scaffolds show better therapeutic activities and strong luminescence properties. By the way, we try to incorporate therapeutic as well as diagnostic properties in a single molecule, which are termed as theranostic molecules (see the below representations) that are explored in the lab.
Research Interest
 |
 |
Front-Cover Page Article in JOC, 2021, 68, 10, 7069-7077 (https://pubs.acs.org/toc/joceah/current) Also see: JOC/OL Tweeter handles Tweet: https://twitter.com/JOC_OL/status/1395766018421764099?s=08 |
- Synthesis of Fused Poly-N-heterocyclic scaffolds by Intramolecular Cyclization via Late-stage C-H modification reactions and Their Efficacy Studies
Our lab is engaged in designing and synthesizing new fused poly-N-heterocyclic molecules using transition metal-catalyzed C-H activation reaction. Strategically, we have used many biologically active heterocycles like quinozolinone, Indole, carbazole, imidazole, etc, as our starting materials. These individual scaffolds have potential therapeutic importance. These fused poly-heterocycles generally showed better therapeutic properties than the individual heterocycles.
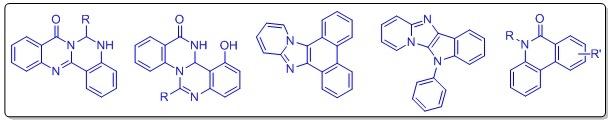
Interestingly, they are usually highly fluorescence active also and show interesting biological properties. Hence structurally important these scaffolds can be well used as biomarkers, utilizing their strong fluorescence properties as well as their new therapeutic efficacies, can also be explored. We have thus further investigated these molecules against different cancer cell lines and tried to develop new therapeutic agents.
Some of the ongoing Research Topics:
- Synthesis of natural products-inspired hybrid scaffolds and their application in therapeutics.
- C-H Activation methodology to construct novel N-Fused Polyheterocyclic molecular scaffolds.
- Design, Synthesis of New pH-dependent Molecular switches & study their properties
- Synthetic methodology using Photo-redox chemistry
- Medicinal Chemistry (New Therapeutics Design) targeting different proteins/enzymes responsible for diseases like Cancer, Neurodegeneration, Covid & SARS-Cov-2 etc
- Design and synthesis of small peptide/peptidomimetics (cyclic) and study their Structural/biophysical/biological properties.
- Design, synthesis, biophysical and biological property studies of dpeptide/ non-peptide based nano-materials.
- Nanoparticle-based drug delivery system using anticancer drugs and de novo synthesized bioactive molecules in the lab.
- Novel Ligand as biomarkers, sensors (Fluoride, Hydrazine, H2O2 etc), Organelle Targeting Fluorophores, OTFPs (Lysosome, Mitochondria, ER, Nucleolus etc) .
Some of the Published Research Activities from the group:
1. Synthesis of N-Fused Triazole–Piperazine–Quinazolinones ‘Hybrid-heterocycle’ via One-Pot Tandem Click Reaction and Cross-Dehydrogenative Coupling
A one pot protocol to synthesize tetracyclic triazole-piperazine-quinazolinone fused N-heterocyclic scaffolds is reported in this paper. In this strategy, a tandem approach of two highly efficient synthetic reactions, Click and Cross dehydrogentive coupling (CDC) reactions, with high atom economy were employed to get the target N-fused scaffolds.
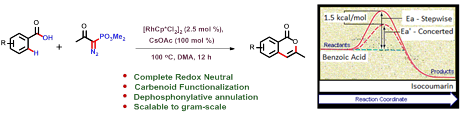
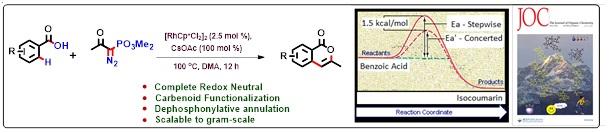
2. Synthetic and Computational Studies on RhIII-Catalyzed Redox Neutral Cascade of Carbenoid Functionalization and Dephosphonylative Annulation.
A Rh(III)-catalyzed regioselective redox-neutral cascade process of carbenoid functionalization followed by dephosphonylative annulation of benzoic acids with α-Diazo-β-keto phosphonatehas been realized, which led to the direct synthesis of privileged 3-substituted isocoumarin scaffold. According to the best of our knowledge, this is the first report of a complete redox neutral method to synthesize isocoumarins using C-H functionalization strategy.
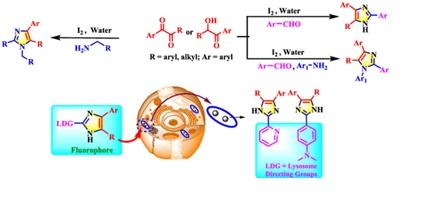
3. Polysubstituted Imidazoles were synthesized in a modular way via Iodine/H2O and their Cell-Imaging studies revealed it as a LysoTracker Molecules.
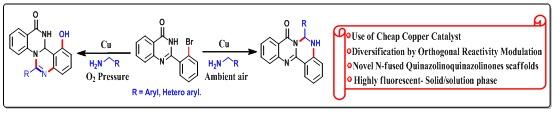
4. Cu-Catalyzed Direct Diversification of 2-(2-Bromophenyl)quinazolin-4(3H)-ones through Orthogonal Reactivity Modulation (Organic Synthesis, OL, 2019) is reported here. A modular strategy to obtain three different products from a single substrate was developed. The present methodology unveils new step-economical and cost-efficient routes to access diverse fused quinazolinoquinazolinone derivatives which are not prevalent in literature.
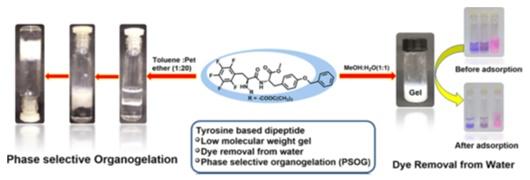
5. Solvent-Assisted Tyrosine-based Dipeptide forms low-Molecular weight gel preparation and Its potential use in dye removal and oil spillage separation from water are reported in this paper (Nano-materials, Acs Omega, 2019).
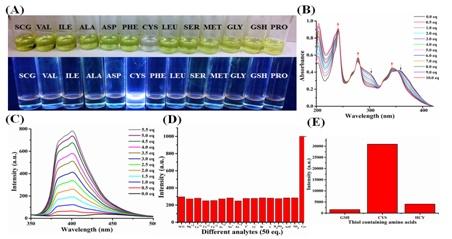
6. An interesting study on the efficient detection of the early events of α-Synuclein aggregation using a Cysteine Specific Hybrid Scaffold (Chemo sensing) is reported in the ‘Biochemistry’ journal. α-Synuclein aggregation is responsible for Parkinson’s disease.
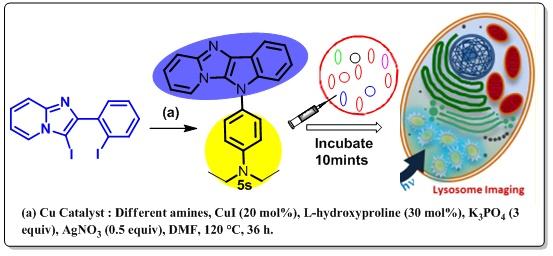
7. A Lysosome tracking agent was developed by a new fluorescent Heterocycle. N-Fused Pyrido–Imidazo–Indole Framework was synthesized via Cu-Catalyzed Tandem N-Arylation reactions. The methodology along with biological results are Reported in our JOC paper.
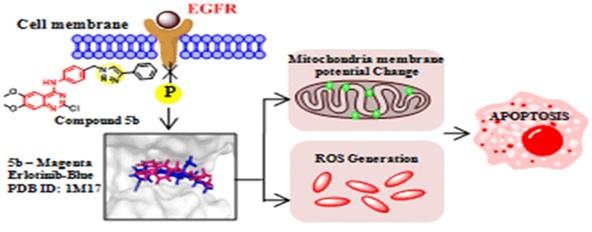
8. Synthesis of Triazole substituted Quinazoline Hybrids compounds were found as anticancer agents. The lead compound was shown to act as EGFR Blocker and ROS inducer.
Credentials
- Chief Scientist, CSIR-IICB, Kolkata, India (2024-onwards)
- Senior Principal Scientist, CSIR-IICB, Kolkata, India
- Principal Scientist, CSIR-IICB, Kolkata, India
- Senior Scientist, CSIR-IICB, Kolkata, India
- Professor, Academy of Scientific and Innovative Research (AcSIR-New Delhi)
- Post Doctoral Research Associate, ICES-A*, Singapore (Prof. K. C.Nicolaou)
- Post Doctoral Research Associate, CRL, Oxford University (Prof. Christopher J. Schofield)
- Ph.D. from Indian Institute of Technology Kanpur, IIT-K, (Mentor: Prof. Javed Iqbal)
- MSc (Organic Chemistry); Calcutta University, Science College, Kolkata, India
- BSc (Organic Chemistry); R.K.M.Vivekananda Centenary (VC) College, Rahara, WB, India
Grants & Supports
1. HSP inhibitors as New Anti-cancer Therapeutics, India, 2016-2019 (Medicinal Chemistry):
Heat shock proteins (Hsps) are over expressed and help cancer cells survive through proper protein-folding. In this project, a simple heterocycle-based scaffold by ring-truncation strategy on a natural product was successfully created. A small library of these scaffolds was synthesized and explored for Hsp-inhibition. Among the library, the lead compound showed cytotoxicity in cervical, ovarian, pancreatic, colon, oral and lung cancers with no cytotoxicity against normal cell. It enhances programed cell death by up-regulating pro-apoptotic and down-regulating anti-apoptotic proteins in two representative ovarian and pancreatic cancer cells. It inhibits Hsp70/Hsp40 leading to the down-regulation of client proteins (Akt/B-raf/Stat3) confirming its role in apoptosis. Binding of the lead molecule with Hsp70 was additionally validated by molecular-modelling. Taken together, this scaffold is a potent Hsp70 inhibitor and its client proteins and therefore, may be considered for the management of cancer.
2. Sickel Cell Anaemia Mission Project, CSIR-India, 2017-2020 (Medicinal Chemistry):
This is one of the very important mission projects, where our group has worked on small molecule Hbs aggregation inhibitors as well as Gardo’s channel inhibitors to combat Sickel Cell anaemia disease, from small molecule therapeutics point of view.
Patents & Publications
Multidisciplinary Publication (2020-present):
- Visible-light-mediated installation of unactivated alkyl groups enabling access to non-natural amino acid derivatives: detailed mechanistic insights; Arindam Manna, Subhankar Shee, Krishnendu Khamaru, Rabindranath Lo and Biswadip Banerji*; Chem. Comm., 2026, asap.
- TBAF Controlled Access to 1,4,5-Trisubstituted and 1,5-Disubstituted Pyrido-triazoles from Tetrazolopyridine with β-Keto Ester and Their Anti-leishmanial Efficacy Studies; Asikul Sk , Prabha Prusti , Arpan Adhikari , Ishita Chatterjee , Susanta Kar , Biswadip Banerji*; J. Org. Chem, 2026, 91, 4, 1686-1693. (https://doi.org/10.1021/acs.joc.5c02708).
- The Visible-Light-Triggered Catalytic Triarylamine EDA Complex Enabled Synthesis of Sulfonylated Quinolines, Pyrroloindoles, and Chromenes by Radical Sulfonylation-Cyclization of Substituted Alkynes with Sulfonyl Chlorides; Kaushik Seal and Biswadip Banerji*, J. Org. Chem, 2026, 91, 4, 1645–1656. (https://doi.org/10.1021/acs.joc.5c02543).
- Isoquinolinone–Naphthoquinone Hybrids as Potent PARP-1 Inhibitors Induce Apoptosis in Glioma via DNA Damage and ROS Generation; Vijay Babu Pathi, Sunny Kumar, Asikul Sk, Subhankar Shee, Mrinal K. Ghosh* and Biswadip Banerji*, J. Med. Chem, 2025, 68, 18, 19153–19168.
- Small Molecule Modulators of α-Synuclein Phase Separation; Debabrata Sarkar, Sumangal Roychowdhury, Rajeev Jain, Krishnananda Chattopadhyay* and Biswadip Banerji*, ACS Chem. Neuro, 2025, 16, 16, 3174–3183
- Access to Pyrido-Pyrimidinone and Imidazopyridine via Fe(Iii)-Mediated Denitrogenative Annulation of Tetrazolopyridine with β-Keto Ester; Asikul Sk, Biswadip Banerji*, Chem. Commun., 2025, 61, 6312-6315 (doi.org/10.1039/D5CC00258C).
- Copper(II)-Mediated Dual Reactivity of 2-(5-Phenylisoxazol-3-yl)aniline: Directed Amination and Oxidative C(═O)─C Cleavage of Amides Enabling Direct Access to Urea Derivatives, Arindam Manna, Krishnendu Khamaru, Vijay Babu Pathi, Subhadip Sett, Prasanta Ghosh, and Biswadip Banerji*, Org. Lett., 2024, 26, 41, 8774–8779. (doi.org/10.1021/acs.orglett.4c03104)
- Hydrazone Based pH-Responsive Configurational Molecular Rotary Switches Containing a New Conjugated π-Electronic Framework; Subhankar Shee, Krishnendu Khamaru and Biswadip Banerji*, Eur. J. Org. Chem., 2024, 27, 1-9 (e202400981).
- Metal-Free Synthesis of N-fused Quinazolino-quinazoline-diones as MALAT1 RNA triple helix Intercalator; Vijay Babu Pathi , Pranotosh Das, Abhyuday Guin, Manish Debnath and Biswadip Banerji*, RSC Med. Chem., 2025,16, 429-434. (doi.org/10.1039/D4MD00614C)
- Crystal to Hydrogel Transformation in S-Benzyl-L-Cysteine-Containing Cyclic Dipeptides - Nanostructure Elucidation and Applications; Saswati Ghosh, Nayim Sepay, Biswadip Banerji*, Chem. Eur. J., 2024, 30, 24, e20240187.
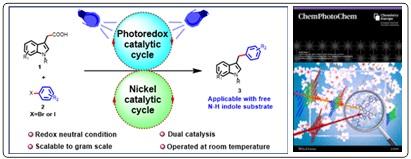
- Merging Photoredox with Nickel Catalysis for Decarboxylative Arylation of Indole-3-Acetic Acids with Aryl Halides; Abhyuday Guin, Suvankar Bera, Vijay Babu Pathi, Biswadip Banerji*; ChemPhotoChem., 2024, 8, e202300338 (doi.org/10.1002/cptc.202300338).
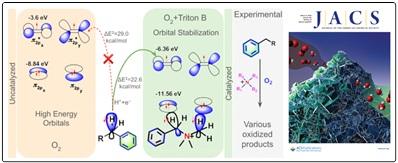
- Metal-Free Activation of Molecular Oxygen by Quaternary Ammonium-Based Ionic Liquid: A Detail Mechanistic Study; Krishnendu Khamaru, Uttam Pal, Rabindranath Lo, Kaushik Seal, Prasanta Ghosh, Nakul Chandra Maiti* and Biswadip Banerji*; J. Am. Chem. Soc. 2024, 146, 10, 6912–6925 (doi.org/10.1021/jacs.3c14366).
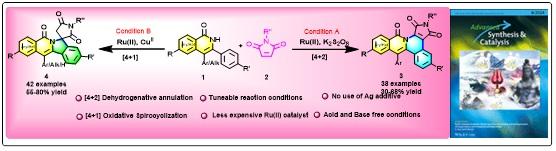
- Ru(II) Catalyzed Oxidative Dehydrogenative Annulation and Spirocyclization of Isoquinolones with N-substituted Maleimides; Kaushik Seal and Biswadip Banerji*; Adv. Synth. Catal., 2024, 366, 8, 1788-1808 (DOI: 10.1002/adsc.202300963).
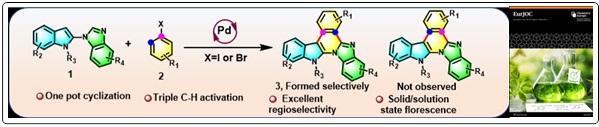
- Pd-Catalyzed Regioselective Domino C-C Bond Formation to Access N-Fused Benzimidazo-Indolo-Isoquinoline Heterocycle; Vijay Babu Pathi, Arindam Manna, Ravuri Srinath, Supriyo Adhikary, Biswadip Banerji*, Eur. J. Org. Chem., 2023, 26, 28, e202300256. (doi.org/10.1002/ejoc.202300256).
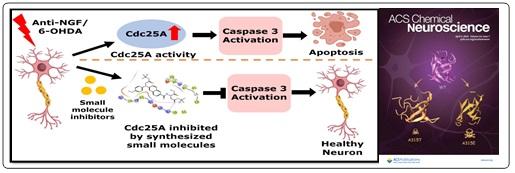
- Small-Molecule Cdc25A Inhibitors Protect Neuronal Cells from Death Evoked by NGF Deprivation and 6-Hydroxydopamine; Pramanik, S.K., Sanphui, P., Das, A.K., Banerji, Biswadip* and Biswas, S.C., ACS Chem. Neuroscience. 2023, 14, 7, 1226–1237.
- Iodine Mediated One-Pot Synthesis of Polyarylated-Oxazoles from Internal Alkynes; Saswati Adhikary and Biswadip Banerji*; ChemistrySelect, 2023, 8, 23, e202300076.
- Cell-imaging studies of highly substituted oxazole derivatives as organelle targeting fluorophores (OTFPs); Adhikary, Saswati; Mukherjee, Kaustuv; Biswadip Banerji*, Scientific Reports. 2022, 12(1), 16555.
- ß-Sheet Induced Helical Self-Assembly Structure Formation by Dityrosine Dipeptide: Crystallographic Evidence and Other Biophysical Studies; Leena Majumder, Nayim Sepay and Biswadip Banerji*; J. Phys. Chem-B, 2022, 126, 28, 5207–5218.
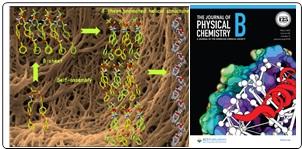
- NMR and vibrational spectroscopic studies on the structure and self-assembly of Two de novo dipeptides in methanol; Leena Mazumdar, Kaushik Bera, Krishnendu Khamaru, Uttam pal, Nakul C. maiti, Biswadip Banerji*, Journal of Molecular Structure, 2022, 1266, 133455.
 S‐Benzyl Cysteine Based Cyclic Dipeptide Super Hydrogelator: Enhancing Efficacy of an Anticancer Drug via Sustainable Release; Ghosh, Saswati, Sayoni Nag, Krishna Das Saha, and Biswadip Banerji*, Journal of Peptide Science. 2022: e340.
S‐Benzyl Cysteine Based Cyclic Dipeptide Super Hydrogelator: Enhancing Efficacy of an Anticancer Drug via Sustainable Release; Ghosh, Saswati, Sayoni Nag, Krishna Das Saha, and Biswadip Banerji*, Journal of Peptide Science. 2022: e340.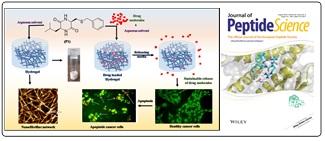 Synthesis of N-Fused Triazole–Piperazine–Quinazolinones via One-Pot Tandem Click Reaction and Cross-Dehydrogenative Coupling: Ravuri Srinath, Arindam Manna, Subhankar Shee, Vijay Babu Pathi, Saswati Ghosh, Krishnendu Khamaru, Nakul Chandra Maiti, and Biswadip Banerji*, Org. lett., https://doi.org/10.1021/acs.orglett.1c03435.
Synthesis of N-Fused Triazole–Piperazine–Quinazolinones via One-Pot Tandem Click Reaction and Cross-Dehydrogenative Coupling: Ravuri Srinath, Arindam Manna, Subhankar Shee, Vijay Babu Pathi, Saswati Ghosh, Krishnendu Khamaru, Nakul Chandra Maiti, and Biswadip Banerji*, Org. lett., https://doi.org/10.1021/acs.orglett.1c03435.
- Synthetic and Computational Studies on RhIII-Catalyzed Redox Neutral Cascade of Carbenoid Functionalization and Dephosphonylative Annulation; Suvankar Bera, Partha Mondal, Debabrata Sarkar, Vijay Babu Pathi, Sourav Pakrashy, Ayan Datta*, Biswadip Banerji*; Journal of Organic Chemistry, 2021, 68, 10, 7069-7077
 Sulphonamide‐Containing Oxazoline Hybrids as New Class of Neuroprotective Agents and Lead Molecule as Autophagy Inducer. Kadaiahgari, C.S., Saha, M., Ravuri, S., Nandigama, S., Saha, K.D. and Biswadip Banerji*, ChemistrySelect, 2021, 6, 5, 1082-1087.
Sulphonamide‐Containing Oxazoline Hybrids as New Class of Neuroprotective Agents and Lead Molecule as Autophagy Inducer. Kadaiahgari, C.S., Saha, M., Ravuri, S., Nandigama, S., Saha, K.D. and Biswadip Banerji*, ChemistrySelect, 2021, 6, 5, 1082-1087.- Use of cyclic peptides to induce crystallization: case study with prolyl hydroxylase domain 2; Rasheduzzaman Chowdhury, Martine I.Abboud, Tom E. McAllister, Biswadip Banerji, Bhaskar Bhushan, John L. Sorensen, Akane Kawamura & Christopher J. Schofeld ; Scientific Reports, 2020, 10, Article number: 21964.
- Polysubstituted Imidazoles as LysoTracker Molecules: Their Synthesis via Iodine/H2O and Cell-Imaging Studies. Saswati Adhikary, Leena Majumder, Sourav Pakrashy, Ravuri Srinath, Kaustuv Mukherjee, Chitra Mandal and Biswadip Banerji*. ACS Omega, 2020, 5, 24, 14394–14407.
Previous:
- Cu-Catalyzed Direct Diversification of 2-(2-Bromophenyl)quinazolin-4(3H)-ones through Orthogonal Reactivity Modulation; Satadru Chatterjee, Ravuri Srinath, Suvankar Bera, Krishnendu Khamaru, Afifa Rahman and Biswadip Banerji*; Org. Lett. 2019, 21, 22, 9028–9032.
- Rh(III) Catalyzed Decarboxylative o-Acylation of Arenes Bearing an Oxidizing Directing Group; Suvankar Bera, K. Chandrasekhar, Satadru Chatterjee, Sunil Kumar Killi, Debabrata Sarkar and Biswadip Banerji*; European Journal of Organic Chemistry, 2019, 24, 3877-3881
- Efficient Detection of Early Events of α-Synuclein Aggregation Using a Cysteine Specific Hybrid Scaffold. Satadru Chatterjee, Sumanta Ghosh, Snehasis Mishra, Krishna Das Saha, Biswadip Banerji*, Krishnananda Chattopadhyay; Biochemistry, 2019, 58, 1109-1119.
- Green Synthetic Approach towards Polyarylated Oxazoles via Iodine‐Catalyzed One‐Pot Sp3 C−H Functionalization in Water: From Natural Product Synthesis To Photophysical Studies; Biswadip Banerji*, Saswati Adhikary, Leena Majumder, Saswati Ghosh; Asian Journal of Organic Chemistry; 2019, 8, 4, 514-525.
- Detection of Lysosome by a Fluorescent Heterocycle: Development of Fused Pyrido–Imidazo–Indole Framework via Cu-Catalyzed Tandem N-Arylation; Biswadip Banerji*, Satadru Chatterjee, Kadaiahgari Chandrasekhar, Saswati Ghosh, Kaustuv Mukherjee, Chitra Mandal; Journal of Organic Chemistry; 2018, 83, 13011-13018.
- Synthesis of Triazole-Substituted Quinazoline Hybrids for Anticancer Activity and a Lead Compound as the EGFR Blocker and ROS Inducer Agent; Biswadip Banerji*, Kadaiahgari Chandrasekhar, Kancham Sreenath, Saheli Roy, Sayoni Nag, Krishna Das Saha; ACS Omega; 2018, 3, 16134-16142.
- Cellular Detection of Hydrazine as Isoniazid Metabolite by a New Turn‐On Fluorescent Probe: Synthesis, Live Cell Imaging and In Vitro Toxicity Studies; Biswadip Banerji*, Chandrasekhar K, Satadru Chatterjee, Sunil Kumar Killi, Chandraday Prodhan, Keya Chaudhuri. Chemistry Select; 2018, 3, 12816-12823.
- Synthesis and DNA-Binding Studies of A New Cyclic Dimeric Symmetrical Pseudo-Turn Mimetic; Biswadip Banerji*, Satadru Chatterjee, Sunil Kumar Killi, D. Srinivas, Chandraday Prodhan, Atul Katarkar, Keya Chaudhuri, ChemistrySelect, 2018, 3, 7, 2103-17.
- A Metal-Free Oxidative Carboannulation Approach towards Synthesis of 2,3-Diarylindenones and Its Regioisomers; Biswadip Banerji*, Leena Majumder, Saswati Adhkaryi, Chemistry Select, 2018, 3, 5, 1381–1384.
- Formation of Annular Protofibrillar Assembly by Cysteine Tripeptide: Unraveling the Interactions with NMR, FTIR, and Molecular Dynamics; Biswadip Banerji*, Moumita Chatterjee, Uttam Pal, and Nakul C. Maiti*; Journal of Physical Chemistry-B, 2017, 121 (26), pp 6367–6379.
- Palladium-Catalyzed Direct Synthesis of Phenanthridones from Benzamides through Tandem N–H/C–H Arylation; Biswadip Banerji*, Satadru Chatterjee, K. Chandrasekhar, Chinmay Nayan, Sunil Kumar Killi; European. Journal of Organic Chemistry, 2017, 35, 5214–5218.
- Expedient synthesis of a phenanthro-imidazo-pyridine fused heteropolynuclear framework via CDC coupling: a new class of luminophores; Biswadip Banerji,* Satadru Chatterjee, K. Organic & Biomolecular Chemistry, 2017, 15, 4130-4134.
- Envisaging Structural Insight of a Terminally Protected Proline Dipeptide by Raman Spectroscopy and Density Functional Theory Analyses; Supriya Das, Uttam Pal, Moumita Chatterjee, Sumit Kumar Pramanik, Biswadip Banerji*, and Nakul C. Maiti*; Journal of Physical. Chemistry-A, 2016, 120 (49), pp 9829–9840.
- Tripeptide consisting of benzyl protected di-cysteine and phenylalanine forms spherical assembly and induces cytotoxicity in cancer cells via apoptosis; Biswadip Banerji*, Moumita Chatterjee, Chandraday Prodhan and Key Chaudhuri, RSC Advances. 2016, 6, 112667-112676.
- Neo-tanshinlactone D-ring modified novel analogues induce apoptosis in human breast cancer cell via DNA damage, Biswadip Banerji*, Sunil Kumar Killi, Atul Katarkar, Satadru Chatterjee, Yellaiah Tangella, Chandraday Prodhan, Keya Chaudhuri; Bioorganic & Medicinal Chemistry, 2017, 1;25(1):202-212.
- Molecular Details of Acetate Binding to a New Diamine Receptor by NMR and FT-IR Analyses, Biswadip Banerji*, Moumita Chatterjee, Uttam Pal, and Nakul Chandra Maiti*, Journal of Physical Chemistry-A, 2016, 120 (15), 2330–2341.
- Regioselective Synthesis of Quinazolinone-/Phenanthridine-Fused Heteropolycycles by Pd-Catalyzed Direct Intramolecular Aerobic Oxidative C-H Amination from Aromatic Strained Amides; Biswadip Banerji*; Suvankar Bera, Satadru Chatterjee, Sunil Kumar Killi, Saswati Adhikary; Chemistry–A European Journal, 2016, 3506-3512.
- Silver-Catalyzed Azide–Alkyne Cycloaddition (AgAAC): Assessing the Mechanism by DFT Calculations, Biswadip Banerji,* K Chandrasekhar, Sunil Kumar Killi, Sumit Kumar Pramanik, UttamPal, Sudeshna Sen and Nakul Chandra Maiti, RSc Open Science.
- Synthesis and cytotoxicity studies of 1-propenyl-1,3-dihydro-benzimidazol-2-one, Biswadip Banerji* and Sumit kumar Pramanik; Journal of Chemical Biology, 2015, 8(3), 73.
- Binding studies of creatinine and urea on iron-nanoparticle; Biswadip Banerji*, Pramanik, Sumit Kumar; SpringerPlus, 4.1 (2015): 1-9.
- Copper (I) oxide nanoparticle and tryptophan as its biological conjugate: a modulation of cytotoxic effects. Mritunjoy Maity, Sumit Kumar Pramanik, Uttam Pal, Biswadip Banerji and Nakul Chandra Maiti; Journal Nanoparticle Research, 2013, 16 (1), 1-13.
- Efficacy of Cyclin dependent kinase 4 (Cdk4) inhibitors as potent neuroprotective agents in cellular models of Alzheimer’s disease. Priyankar Sanphui, Sumit kumar Pramanik, P. Moorthi, Biswadip Banerji* and Subhas C Biswas; PLOS ONE, 2013, 8 (11), e78842.
- Synthesis of Sulfonamide Derived from New Tetrahydroquinazoline Derivatives via Intramolecular cyclization. Biswadip Banerji*, Moumita Chatterjee, Suvankar Bera and Mohd Usman Mohd Siddique; JMED Research, 2014, Article ID 464776, DOI: 10.5171/2014.464776.
- Synthesis and Cytotoxicity Studies of Novel Triazolobenzoxazepine as New Anticancer Agents Biswadip Banerji*, Sumit Kumar Pramanik , Priyankar Sanphui, Sameer Nikhar, and Subhas Chandra Biswas; Chemical Biology & Drug Design, 2013, 82 (4), 401-409.
- Potent anticancer activity of cystine-based dipeptides and their interaction with serum albumins, Biswadip Banerji*, Sumit Kumar Pramanik , Uttam Pal and Nakul Chandra Maiti; Chemistry Central Journal, 2013, 7:9
- Dipeptide derived from benzylcystine forms unbranched nanotubes in aqueous solution, Biswadip Banerji*, Sumit Kumar Pramanik , Uttam Pal and Nakul Chandra Maiti Journal of Nanostructure in Chemistry, 2013, 3 (1), 12
- Conformation and Cytotoxicity of a Tetrapeptide Constellated with Alternative D- and L Proline, Biswadip Banerji, Sumit Kumar Pramanik , Uttam Pal and Nakul Chandra Maiti RSC Advance., 2012, 2, 6744-6747
- Synthesis, characterization and cytotoxicity study of magnetic (Fe3O4) nanoparticles and their drug conjugate Biswadip Banerji, Sumit Kumar Pramanik , Samir Mandal , Nakul Chandra Maiti and Keya Chaudhuri RSC Advance., 2012, 2, 2493-2497.
- Total Synthesis of the Originally Proposed and Revised Structure of Palmerolide A; K. C. Nicolaou*, Ramakrishna Guduru, Ya-Ping Sun, Biswadip Banerji, David Chen Yu- Kai* Angew. Chem. Int. Ed. 2007, 46, 5896.
- Total Synthesis of the Proposed and Revised Structure of Palmerolide A; K. C. Nicolaou*, Ya-Ping Sun, Ramakrishna Guduru, Biswadip Banerji, David Chen Yu- Kai* J. Am. Chem. Soc, 2008, 130(11), 3633-3644.
- Selective Inhibition of Factor Inhibiting Hypoxia Inducible Factor Michael A. McDonough, Luke A. McNeill, Melanie Tilliet, Cyril A. Papamicaël, Qiu-Yun Chen, Biswadip Banerji, Kirsty S. Hewiston, Christopher J. Schofield* J. Am. Chem. Soc, 2005, 127(21), 7680-7681.
- The Inhibition of Factor Inhibiting Hypoxia Inducible Factor (FIH) by β-oxocarboxylic acids; Biswadip Banerji, Ana Conejo-Garcia, Luke McNeill, Michael A. McDonough, Matthew Buck, Kirsty S. Hewitson, Neil J. Oldham & Christopher J. Schofield*; Chem.Com, 2005, 43, 5438.
- Design, Synthesis and Conformational analysis of Inverse γ-Turn Cyclic Peptidomimetic of Hypoxia Inducible Factor (HIF); MorakotKaewpet, Barbara Odell, Michael King, Biswadip Banerji, , Christopher J. Schofield* & Timothy D. Claridge; Org. Biomol. Chem, 2008, 6, 3476.
- Palladium (0) CatalyzedRegioselective Synthesis of α-Dehydro-β-Amino Ester From Amines and Allyl acetates: Synthesis of α-Dehydro-β Aminoacid Derived Cyclic Peptide As a Constrained β-turn Mimic; S. Rajesh, Biswadip Banerji and Javed Iqbal*; J. Org. Chem, 2002, 67(22), 7852-7857.
- Synthesis of Cyclic Pseudo 310-Helical Structure From β-Amino Acid-L-Proline Derived Tripeptides Via Ring Closing Metathesis; Biswadip Banerji, B. M. Mallesh, S. Kirankumar, A.C. Kunwar and JavedIqbal.*; Tetrahedron Lett., 2002, 43(36), 6479-6483.Synthesis of Cyclic β- Turn Mimics From L-Pro-Phe / Phe-L-Pro Derived di- and Tri-peptides Via Ring Closing Metathesis: The Role of Chirality of Phe- Residue During Cyclisation; Biswadip Banerji, Madhushree Bhattacharya, Rajesh B. M, Saibal Kumar Das and Javed Iqbal.*; Tetrahedron Lett., 2002, 43(36), 6473-6477.
- A Facile Conversion of 310 Helical Structure To Cyclic β-Turn Mimic In Dehydro- Phenylalanine Derived Small Peptides Through Ring Closing Metathesis T. V. R. S. Shastry, Biswadip Banerji, S. Kiran Kumar, A. C. Kunwar, J. P. Nandy and Javed Iqbal*; Tetrahedron Lett., 2002, 43(42), 7621-7625.
- Synthesis of β-Methyl-β-alanine-L-Proline-XAA Tripeptides by Yb(OTf)3 Catalysed Michael Addition of Amines To N-Crotonyl-L-Proline-XAA: A Versatile Route To Cyclic β-Methyl-β-alanine Derived Tripeptides Via Ring-Closing Metathesis; BiswajitSaha, Debasis Das, Biswadip Banerji and Javed Iqbal*; Tetrahedron Lett., 2002, 43(36), 6467-6471.
- Synthesis of Small Cyclic Peptides By Intramolecular Heck Reaction; V. Balraju, Rajmohan Reddy, G. R. Madhavan, Biswadip Banerji and Javed Iqbal* Tetrahedron Lett., 2003, 44(2), 353-356.
- Synthesis of an α-Dehydro-β-Amino Acid Derived Cyclic Peptide As a Constrained β-Turn Mimic S. Rajesh, J. Shrivastava, Biswadip Banerji and Javed Iqbal*; ARKIVOC, 2001, 2, 10, 20-26.
- a-Dehydro-β-Aminoacid Derivatives As Turn Inducers: Synthesis of Potential HIV Protease Inhibitors Based On Structural Mimicry S. Rajesh, J. Srivastava, Biswadip Banerji and Javed Iqbal*, Ind. J. Chem, 2001, 40B, 1029-1032.
- Synthesis of L-proline derived cyclic β-turn mimics via ring closing metathesis Rajesh B. M, M. Bhattacharya, Biswadip Banerji and Javed Iqbal*; ARKIVOC, 2004, (viii),111.
